“IM [or intramuscular] injections yielded expression at the injection site (68%), liver (12%), and kidneys (11%)” (2).
“Given this innovative vaccine platform, we examined the bio-distribution of the mRNA vaccines for both routes of administration [in male rats] […] Following IM administration, the maximum concentration (Cmax) of the injection site muscle was 5,680 ng/mL, and the level declined with an estimated t1/2 of 18.8 hr. Proximal lymph nodes had the second highest concentration at 2,120 ng/mL[…], suggesting that [the] mRNA distributes from the injection site to systemic circulation through the lymphatic system. The spleen and liver had a mean Cmax of 86.9 ng/mL […] and 47.2 ng/mL […], respectively. In the remaining tissues and plasma, H10 mRNA was found at 100- to1,000-fold lower levels” (3).
“while mRNA mainly transfects cells near the injection site it could hypothetically reach any cell in the body (Maugeri et al., 2019; Pardi et al., 2015)” (4).
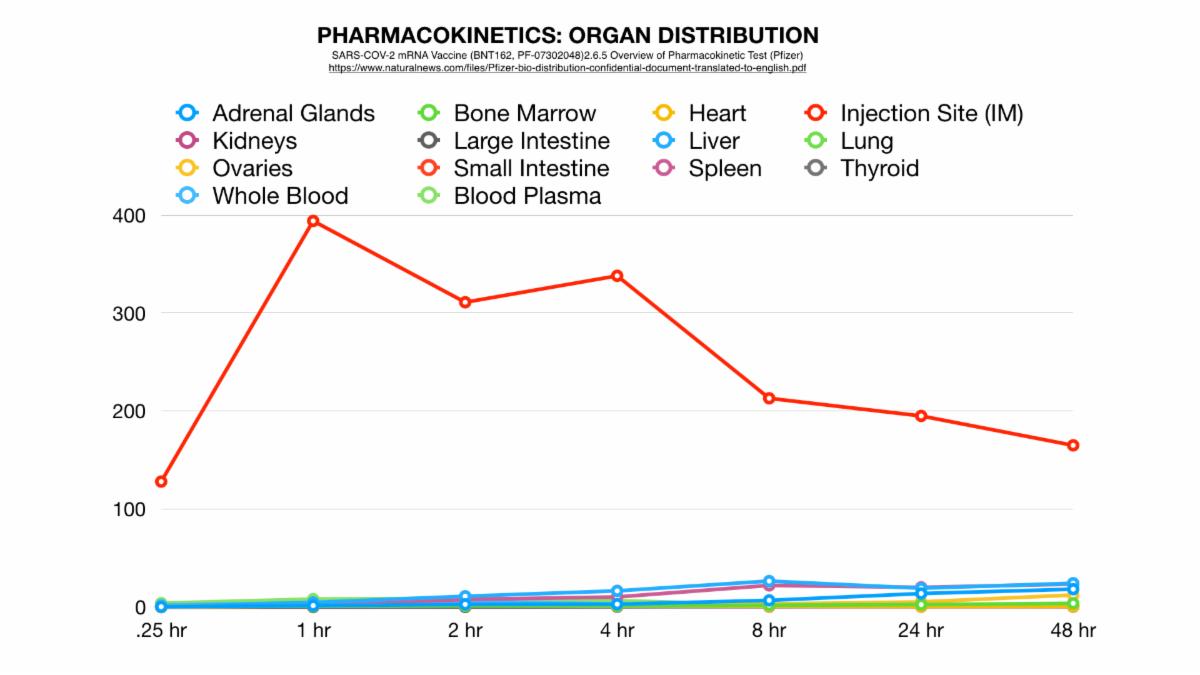
To sum up these findings, as in other lipid nanoparticle studies featuring the intramuscular injection route, the majority of the vaccine does in fact reside near the injection site, however, to a lesser yet potentially significant extent these substances can migrate to the liver, spleen, adrenal glands, and other organs. How do we know this? Partly because of the larger body of evidence we’ve just discussed, but also specific insight from a study shared by Pfizer at the behest of the Japanese government (1).
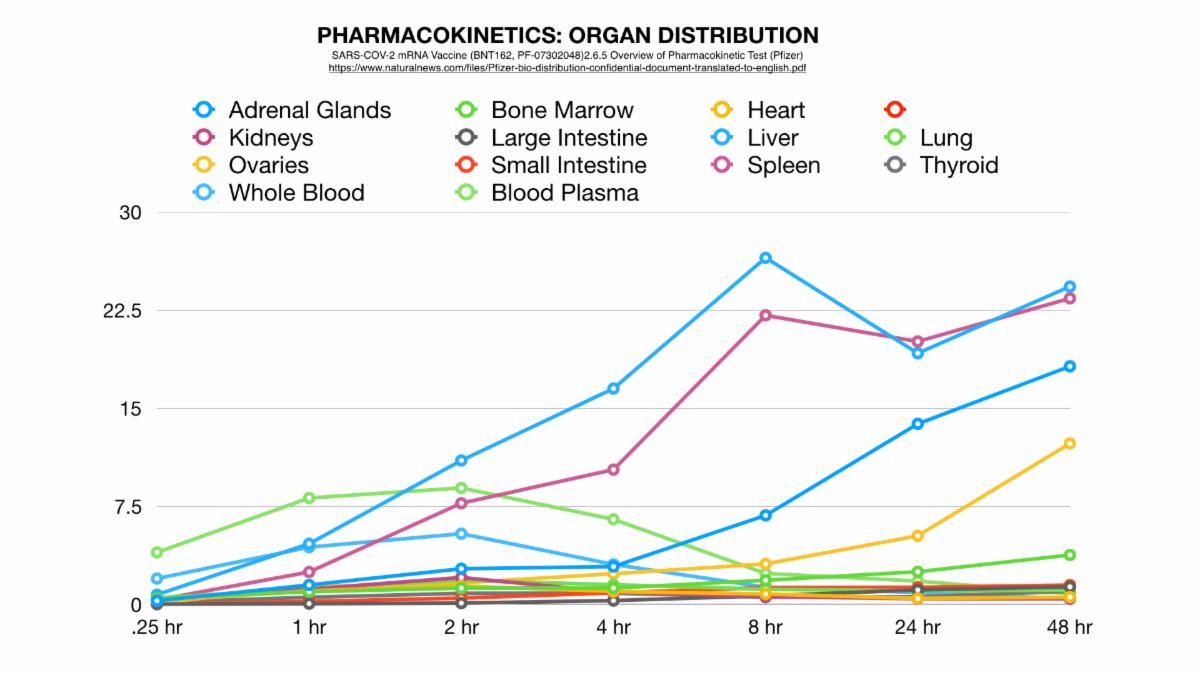
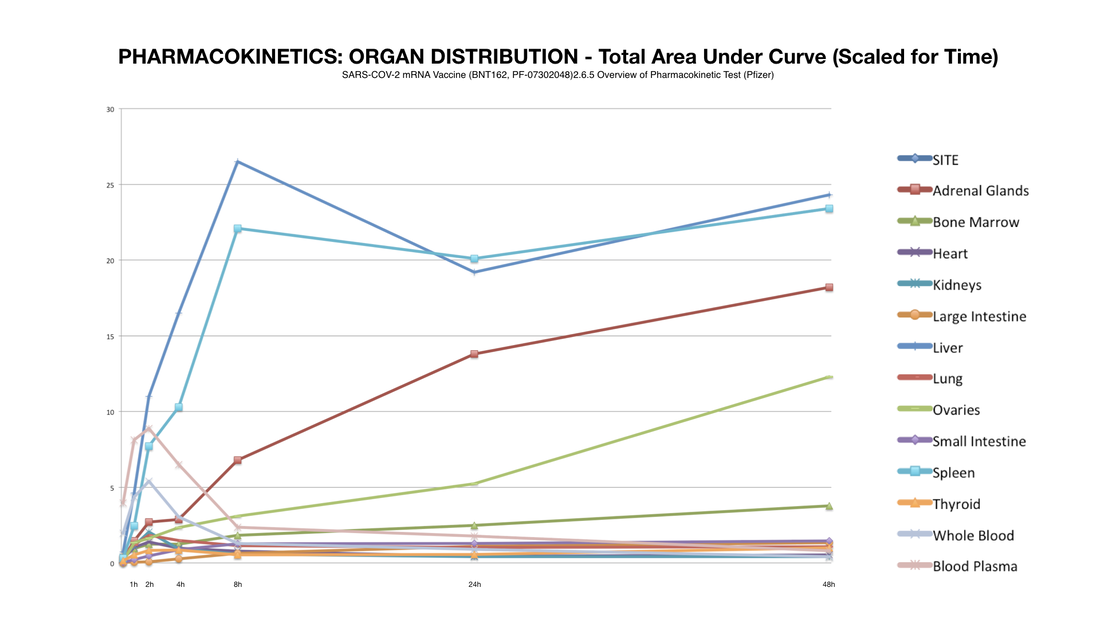
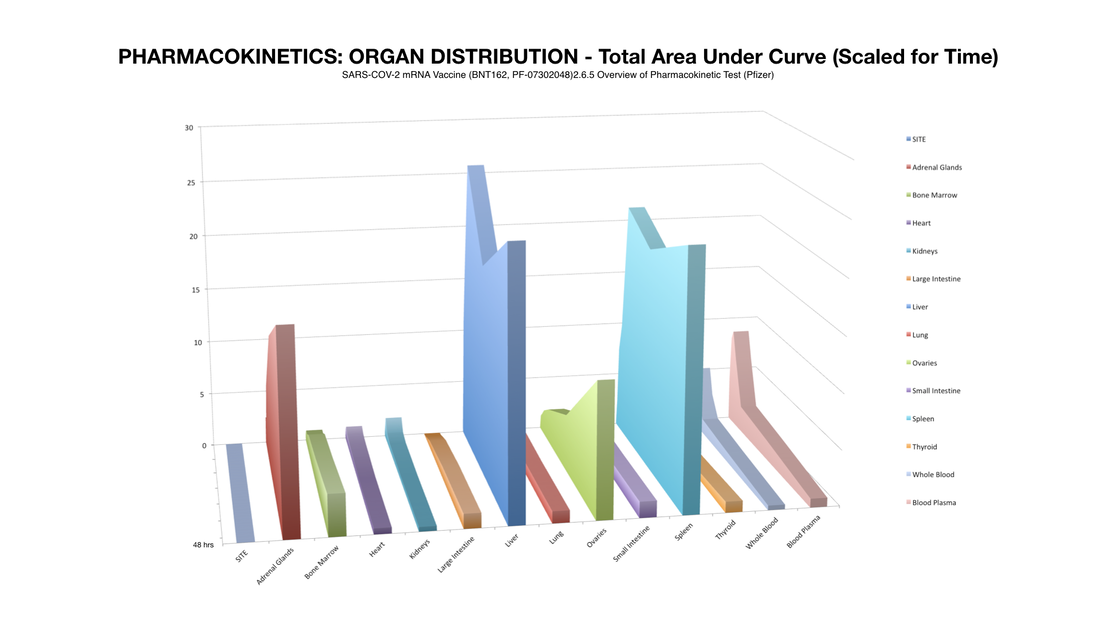
Here's what they disclosed: Figure A shows all significant distribution sites plotted, Figure B shows distribution curves without reference to the injection site (to get a better view of the sites of lesser concentration).
So, if we could see the data post 48 hours we would expect more of this potentially dangerous trend. Potentially most disturbing is that by this time an alarming percentage of LNP’s had made their way to the most vital of the female reproductive organs: the ovaries. This is a strange occurrence considering that these organs are not a site of massive blood perfusion like the spleen or liver. To make matters worse, this increase follows an exponential curve: a trend that could lead to tremendously increasing concentration of nanoparticles in these specific organs in a much shorter amount of time as previously shown.
A study in the scientific journal Controlled Release, sheds some further insight into this phenomenon In fact, researchers found that, over time, nanoparticle injections do lead to inordinate accumulation in the ovaries:
"Several nanocarrier systems are frequently used in modern pharmaceutical therapies. Within this study a potential toxicity risk of all nanoscaled drug delivery systems was found. An accumulation of several structurally different nanocarriers but not of soluble polymers was detected in rodent ovaries after intravenous (i.v.) administration. Studies in different mouse species and Wistar rats were conducted and a high local accumulation of nanoparticles, nanocapsules and nanoemulsions in specific locations of the ovaries was found in all animals” (10).
Furthermore, these researchers issued a warning that interventions utilizing nano-technology for drug/ vaccine delivery should not be approved for use on females before results of long-term studies could be analyzed:
"The findings of this study emphasise[sic] the role of early and comprehensive in vivo studies in pharmaceutical research” (ibid).
Having established the fact of: #1) the potential for systemic spread of mRNA vaccines; and, #2) the ovaries may receive a disproportionate amount of the payload, the next important question would be: what effect do the lipid nanoparticles and spike protein mRNA have on the ovaries (and, for that matter, other tissues as well)?
First of all, lipid nanoparticles themselves show ability to disrupt cellular integrity by triggering inflammation:
“Here we show that the LNPs used for many preclinical studies are highly inflammatory” (4).
Secondly, the actual transfection of affected cells could have consequences leading to tissue destruction:
“[After transfection of a cell with Spike Protein mRNA] the resulting translated protein could be presented on MHC-I in the form of peptides or displayed as a whole protein in the cell membrane. In both cases, cells with the vaccine peptide/protein on their surfaces could be targeted and killed by cells of the adaptive and innate immune system” (ibid).
In other words, every cell that is “transfected” by the Covid-19 spike protein-producing mRNA would be a potential target for immune-mediated cellular destruction. Additional vaccine boosters would increase this risk dramatically, as well as prior Covid-19 infection, due to the fact that antibodies and immune memory against the spike protein would intensify the attack on cells expressing this whole protein or subsequent segments in MHC-1.
Additional risks could potentially stem from this physiologic phenomenon: initiation of autoimmune disease, immune cell programming issues dealing with self-recognition, stem cell abnormalities, spontaneous abortion of pre uterine-implanted embryos, and many more.
Furthermore, organs and tissues not normally at risk by an actual SARS-CoV-2 viral infection could be put in harms way. Whereas the latter needs certain receptors to infect a cell (ACE II) lipid nanoparticles could potentially be absorbed into almost any cell in the body (5, 11).
The implications of this happening on a population-wide scale are extremely troubling. To make matters worse, there are currently no studies showing the confounding effect of other variables: such as physiologic factors affecting pharmacokinetics of the vaccine. For one, the massive population based public health measures such as the hypercapnia (elevated CO2) associated with prolonged occlusive masking (12). Studies indicate that the corresponding effect upon a compound known as NADPH oxidase could decrease the oxidation of LNP’s, in this case ALC-0315, the lipid encasing the spike protein mRNA- potentially slowing excretion/ elimination and increasing systemic spread (13, 14).
Another factor would be increased vascularity occurring at the injection site (deltoid muscle) among athletes that would greatly increase systemic spread (15). This phenomenon could be part of the underlying basis for the increase risk of myocarditis seen among healthy athletic individuals following vaccination (16).
Although much is still to be ascertained regarding the potential long term side effects of these novel mRNA vaccines, two things are certain: #1) the shot does not simply stay in the arm (although we still don't know exactly how it translocates to distal organs and tissues or what damage it causes during this process); and #2) the inherent inflammatory and immune-mediated cytotoxicity give a number of additional reasons to be concerned. Can you really blame me for being Covid-19 "vaccine hesitant?"
Healthy Regards,
Ron Meinhardt
Executive Director
Entering Wedge Media
www.enteringwedgemedia.com
P.S.If you havent already seen our eye-opening COVID-19 interview with Dr Jim Meehan click here to watch and share!
References:
1) https://www.pmda.go.jp/drugs/2021/P20210212001/672212000_30300AMX00231_I100_1.pdf
2) Hajj KA, Melamed JR, Chaudhary N, Lamson NG, Ball RL, Yerneni SS, Whitehead KA. A Potent Branched-Tail Lipid Nanoparticle Enables Multiplexed mRNA Delivery and Gene Editing In Vivo. Nano Lett. 2020 Jul 8;20(7):5167-5175. doi: 10.1021/acs.nanolett.0c00596. Epub 2020 Jun 9. PMID: 32496069; PMCID: PMC7781386.
3) Bahl K, Senn JJ, Yuzhakov O, Bulychev A, Brito LA, Hassett KJ, Laska ME, Smith M, Almarsson Ö, Thompson J, Ribeiro AM, Watson M, Zaks T, Ciaramella G. Preclinical and Clinical Demonstration of Immunogenicity by mRNA Vaccines against H10N8 and H7N9 Influenza Viruses. Mol Ther. 2017 Jun 7;25(6):1316-1327. doi: 10.1016/j.ymthe.2017.03.035. Epub 2017 Apr 27. PMID: 28457665; PMCID: PMC5475249.
4) Ndeupen S, Qin Z, Jacobsen S, Estanbouli H, Bouteau A, Igyártó BZ. The mRNA-LNP platform's lipid nanoparticle component used in preclinical vaccine studies is highly inflammatory. Preprint. bioRxiv. 2021;2021.03.04.430128. Published 2021 Jul 23. doi:10.1101/2021.03.04.430128
5) Ryan Cross (2021). "Without These Lipid Shells, There Would Be No mRNA Vaccines for COVID-19". Chemical & Engineering News: 16–19. doi:10.47287/cen-09908-feature1.
6) Katiyar, N., Raju, G., Madhusudanan, P. et al.Neuronal delivery of nanoparticles via nerve fibres in the skin.Sci Rep11, 2566 (2021). https://doi.org/10.1038/s41598-021-81995-x
7) Ahmad E, Feng Y, Qi J, Fan W, Ma Y, He H, Xia F, Dong X, Zhao W, Lu Y, Wu W. Evidence of nose-to-brain delivery of nanoemulsions: cargoes but not vehicles. Nanoscale. 2017 Jan 19;9(3):1174-1183. doi: 10.1039/c6nr07581a. PMID: 28009915.
8) Mistry A, Stolnik S, Illum L. Nanoparticles for direct nose-to-brain delivery of drugs. Int J Pharm. 2009 Sep 8;379(1):146-57. doi: 10.1016/j.ijpharm.2009.06.019. Epub 2009 Jun 23. PMID: 19555750.
9) Suk JS, Xu Q, Kim N, Hanes J, Ensign LM. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv Drug Deliv Rev. 2016;99(Pt A):28-51. doi:10.1016/j.addr.2015.09.012
10) Andreas Schädlich, Stefan Hoffmann, Thomas Mueller, Henrike Caysa, Cornelia Rose, Achim Göpferich, Jun Li, Judith Kuntsche, Karsten Mäder, Accumulation of nanocarriers in the ovary: A neglected toxicity risk?, Journal of Controlled Release, Volume 160, Issue 1, 2012, Pages 105-112, ISSN 0168-3659
11) Liu M, Wang T, Zhou Y, Zhao Y, Zhang Y, Li J. Potential Role of ACE2 in Coronavirus Disease 2019 (COVID-19) Prevention and Management. J Transl Int Med. 2020 May 9;8(1):9-19. doi: 10.2478/jtim-2020-0003. PMID: 32435607; PMCID: PMC7227161.
12) Roberge RJ, Coca A, Williams WJ, Powell JB, Palmiero AJ. Physiological impact of the N95 filtering facepiece respirator on healthcare workers. Respir Care. 2010 May;55(5):569-77. PMID: 20420727.
13) Kogan AKh, Grachev SV, Eliseeva SV, Bolevich S. Uglekislyĭ gaz--universal'nyĭ ingibitor generatsii aktivnykh form kisloroda kletkami (k rasshifrovke odnoĭ zagadki évoliutsii) [Carbon dioxide--a universal inhibitor of the generation of active oxygen forms by cells (deciphering one enigma of evolution)]. Izv Akad Nauk Ser Biol. 1997 Mar-Apr;(2):204-17. Russian. PMID: 9190222.
14) Vlasova II, Kapralov AA, Michael ZP, Burkert SC, Shurin MR, Star A, Shvedova AA, Kagan VE. Enzymatic oxidative biodegradation of nanoparticles: Mechanisms, significance and applications. Toxicol Appl Pharmacol. 2016 May 15;299:58-69. doi: 10.1016/j.taap.2016.01.002. Epub 2016 Jan 6. PMID: 26768553; PMCID: PMC4811710.
15) Jensen L, Bangsbo J, Hellsten Y. Effect of high intensity training on capillarization and presence of angiogenic factors in human skeletal muscle. J Physiol. 2004 Jun 1;557(Pt 2):571-82. doi: 10.1113/jphysiol.2003.057711. Epub 2004 Mar 12. PMID: 15020701; PMCID: PMC1665084.
16) Hudson B, Mantooth R, DeLaney M. Myocarditis and pericarditis after vaccination for COVID-19. J Am Coll Emerg Physicians Open. 2021 Jul 26;2(4):e12498. doi: 10.1002/emp2.12498. PMID: 34337595; PMCID: PMC8313036.

 RSS Feed
RSS Feed